Advanced Microscopy
About us
The Florey Microscopy Facility supports in-house scientists and visitors in using advanced light microscopy methods for their research.
The facility houses a collection of state-of-the-art microscopy equipment and image processing tools. The experienced microscopy team are also available to assist users throughout the entire process of microscopic imaging.
A joint Florey and University of Melbourne facility, the facility regularly organises in-house and international courses to teach basic and advanced light microscopy methods.
Bookings
Researchers from The Florey, neighbouring research institutes, universities and external laboratories are highly encouraged to make use of the systems and equipment within the facility. External users or those new to the facility are encouraged to contact Microscopy staff to discuss access.
Hours of use for external users – Monday to Friday 8 am to 4 pm.
Online booking platformMicroscopes
All users of microscopy equipment need to be trained or demonstrate competence before access is permitted. New users of the facility will need to contact the facility team to arrange access.
Access can be arranged for a variety of state-of-art microscopes and equipment within the facility including:
- Brightfield and multi-channel fluorescence microscopy
- Laser scanning confocal microscopy
- Spinning disk confocal imaging for imaging tissues
- Super resolution imaging using Airyscan 2 and DeepSIM
- Live cell imaging capabilities
- Stereology for quantifying cell numbers and volume
- Neurolucida for neuron structure analysis
- Huygens core and Zen deconvolution
- Analysis tools
Technical specifications
The Florey has seven state-of-art microscopes to assist with and quantify many aspects of your sample – from volume of structures of interest to number of neurons, synaptic contacts, neurite lengths and connectivity to co-localisation and dynamic processes in live cells.
Learn moreServices
Staff within the facility are available to assist users throughout the entire process of microscopic imaging, with experience in:
- experiment planning
- sample preparation
- microscope selection and usage
- image processing and visualisation
- ongoing advice and support for imaging projects.

Analysis
The facility provides access to various software packages to facilitate quantitative analysis of microscope data sets. These programs are installed on a dedicated machine that can be accessed remotely. Please contact the facility staff for details.
Imaris
Imaris is available on the Precision Analysis Workstation, providing 3D or 4D visualisation. Imaris contains step-by-step wizards for analysis such as object detection and Filament Tracer for automatically detecting neurons. This software also includes:
- analysis of spines and other filamentous structures in 2D,3D or 4D datasets
- measurements
- object tracking
- cell measurements
- plotting
- group comparisons with statistical tests
- two-way interface for customization in Matlab, Java or Python.
Huygens HRM and Essential
The facility provides server-based deconvolution using Huygens deconvolution software (SVI) to increase resolution and contrast, decrease noise and correct artefacts such as chromatic shifts, crosstalk and object movement. This program is available on the Offline Image Analysis workstation. Contact the microscopy team for access.
Stereo Investigator and Neurolucida 360 Lite
Stereo Investigator enables researchers to get accurate and reliable measures of total cell number, length, area and volume of cells and structures in tissue samples.
Neurolucida 360 lite can be used to quickly and accurately reconstruct neurons from a variety of labelling and microscopy techniques.
Zen Blue
The Zen Blue software is available on the Precision Analysis Workstation. The program allows researchers to work with .czi files, including post-processing of airyscan data. The package also hosts the Deconvolution Toolkit enabling image restoration via constrained iterative methods and creation of PSF.
FIJI
FIJI is a free image processing package – a ‘batteries-included’ distribution of ImageJ, bundling a lot of plug-ins that facilitate scientific image analysis.
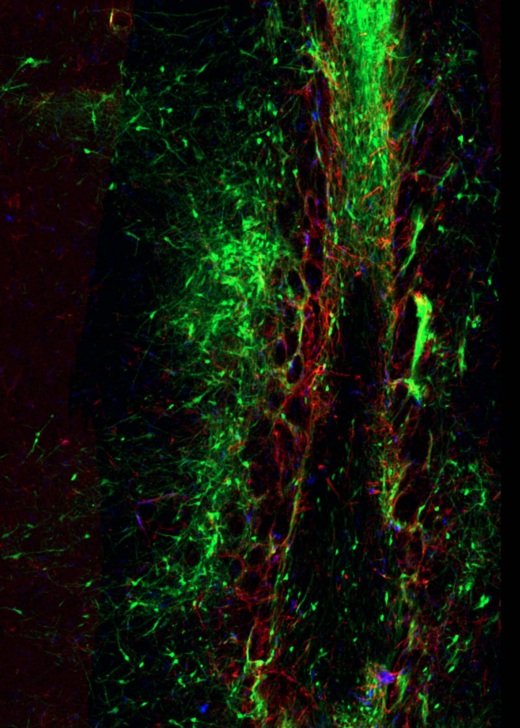
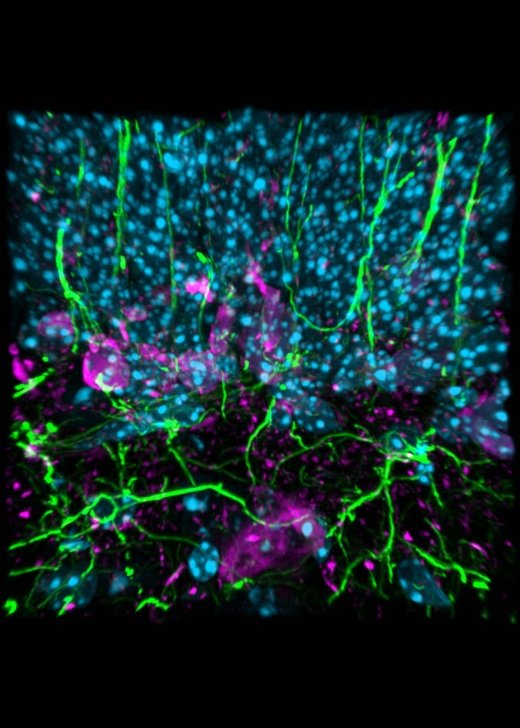
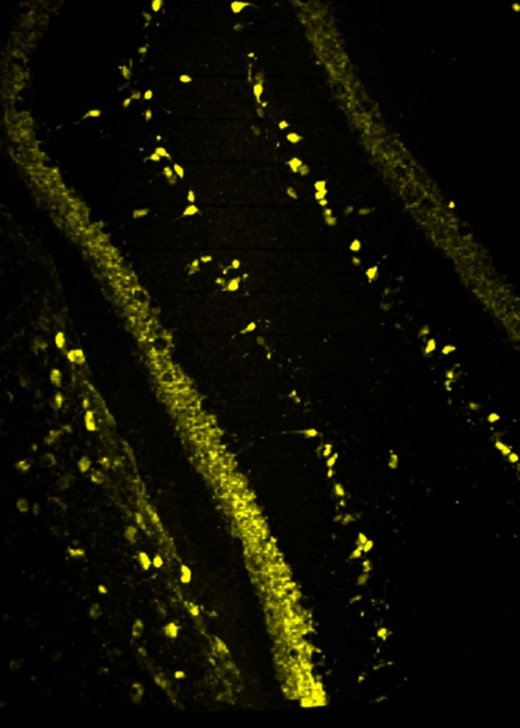
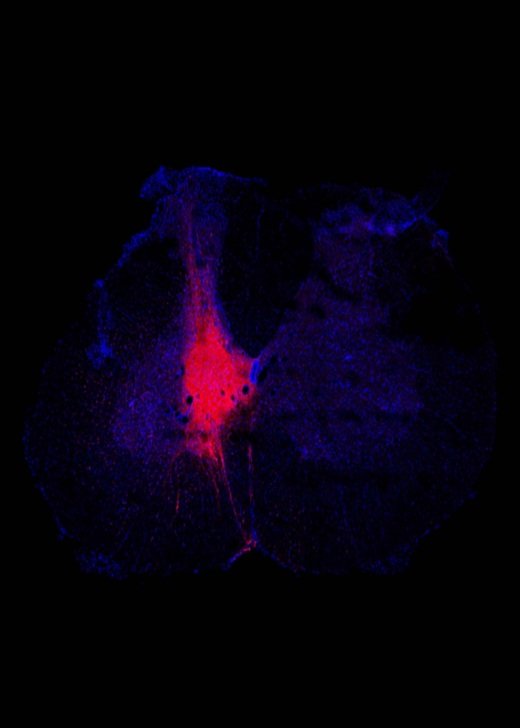
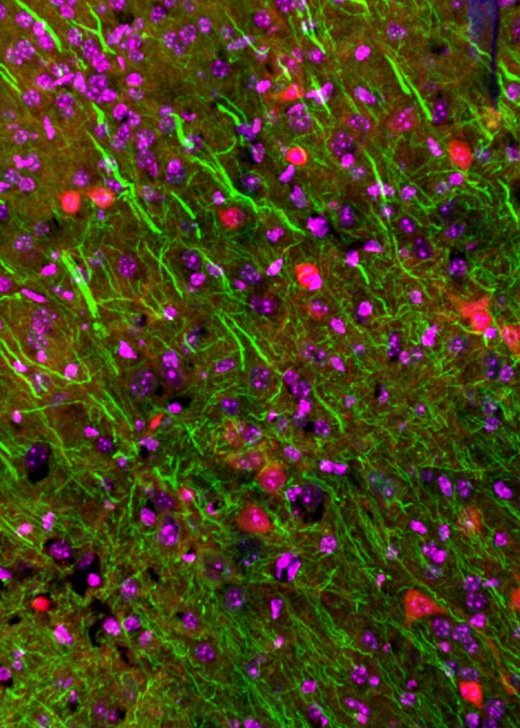
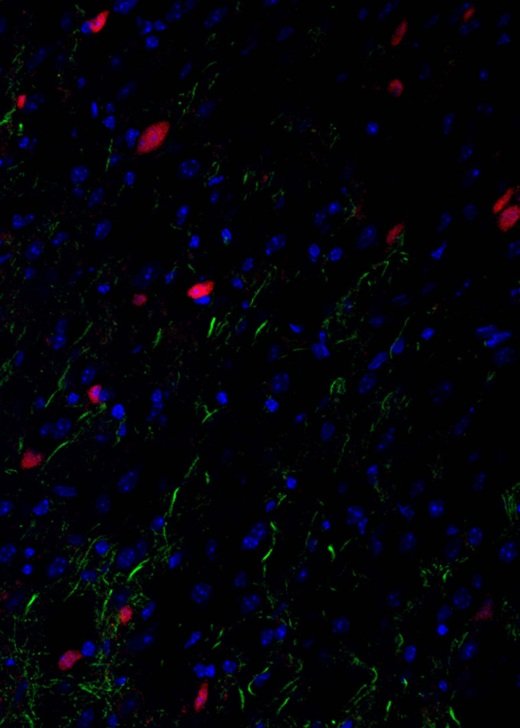
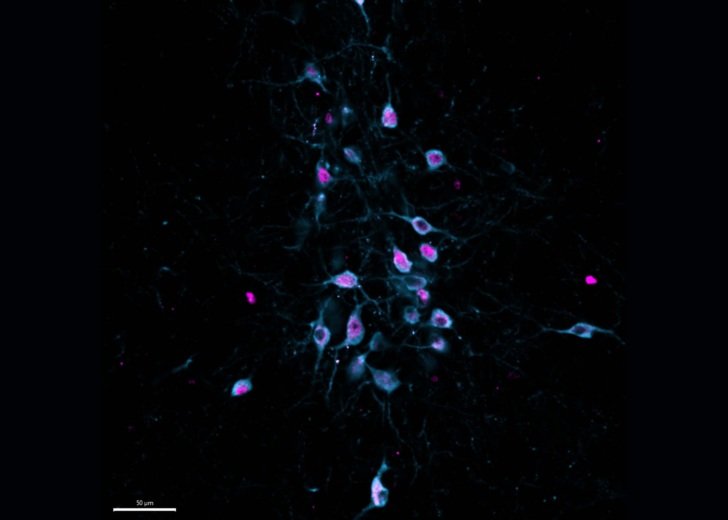
Gallery
Explore some of the microscopic images produced within the facility.
Contact
Microscopy Facility
The Florey
Kenneth Myer Building
30 Royal Parade (corner of Genetics Lane)
Parkville Victoria 3010
Facility staff
Dr Carolina Chavez, Microscopy Facility Manager
Dr Alita Soch, Microscopy Facility Officer