In a world first, Austin Health clinicians have implanted an electrical nerve stimulation device into a patient with Crohn’s disease to prevent inflammation recurring after surgery.
The device, developed and manufactured by the Bionics Institute in collaboration with researchers at The Florey and The University of Melbourne, could put Crohn’s sufferers
into long-term remission.
Crohn’s disease is a condition that causes inflammation to the lining of the gut. Most patients with Crohn’s eventually require bowel surgery to remove sections of gut
damaged by inflammation. However, the disease frequently returns, requiring further surgery.
Crohn’s sufferers are also often at the mercy of life-long drug therapies to manage symptoms.
Co-investigator and team leader Professor John Furness, head of The Florey’s Digestive Physiology and Nutrition Laboratories, said this breakthrough therapy could transform the lives of people suffering from Crohn’s disease.
By stimulating anti-inflammatory nerves and addressing inflammation early, the recurrent inflammation and associated scar tissue development can hopefully be prevented altogether, meaning patients will avoid the cycle of surgery and recovery.
Associate Professor Peter De Cruz, Austin Health’s Director of Inflammatory Bowel Disease Service said Crohn’s disease can have significant impacts on the lives of Australians throughout some of the best years of their lives.
“Australia has among the highest incidence of Crohn’s disease in the world, and affects young people in their 20s to 30s at a time when they are trying to study, enter the workforce or commence families.”
Professor James Fallon, CTO and Head of Research at the Bionics Institute leads the team that developed the groundbreaking device, which was this month implanted onto the vagus nerve of Crohn’s patient, Anthony Becker.
Mr Becker has suffered from Crohn’s disease for as long as he can remember, now into his 40s and with a young family, he is determined to tackle the disease head-on to ensure he maintains a quality of life that is free of surgeries.
Anthony was the first patient to receive the stimulation device earlier this month, and despite being on medication for a number of years, he still required surgery.
If this treatment proves successful, he may never need additional surgical treatments to keep his Crohn’s disease at bay.
Professor James Fallon said: “The vagus nerve controls many functions in the body, such as digestion, heart rate and the immune system. The device is made up of tiny electrodes that stimulate the vagus nerve to trigger the body’s natural defences and prevent inflammation from damaging the gut.
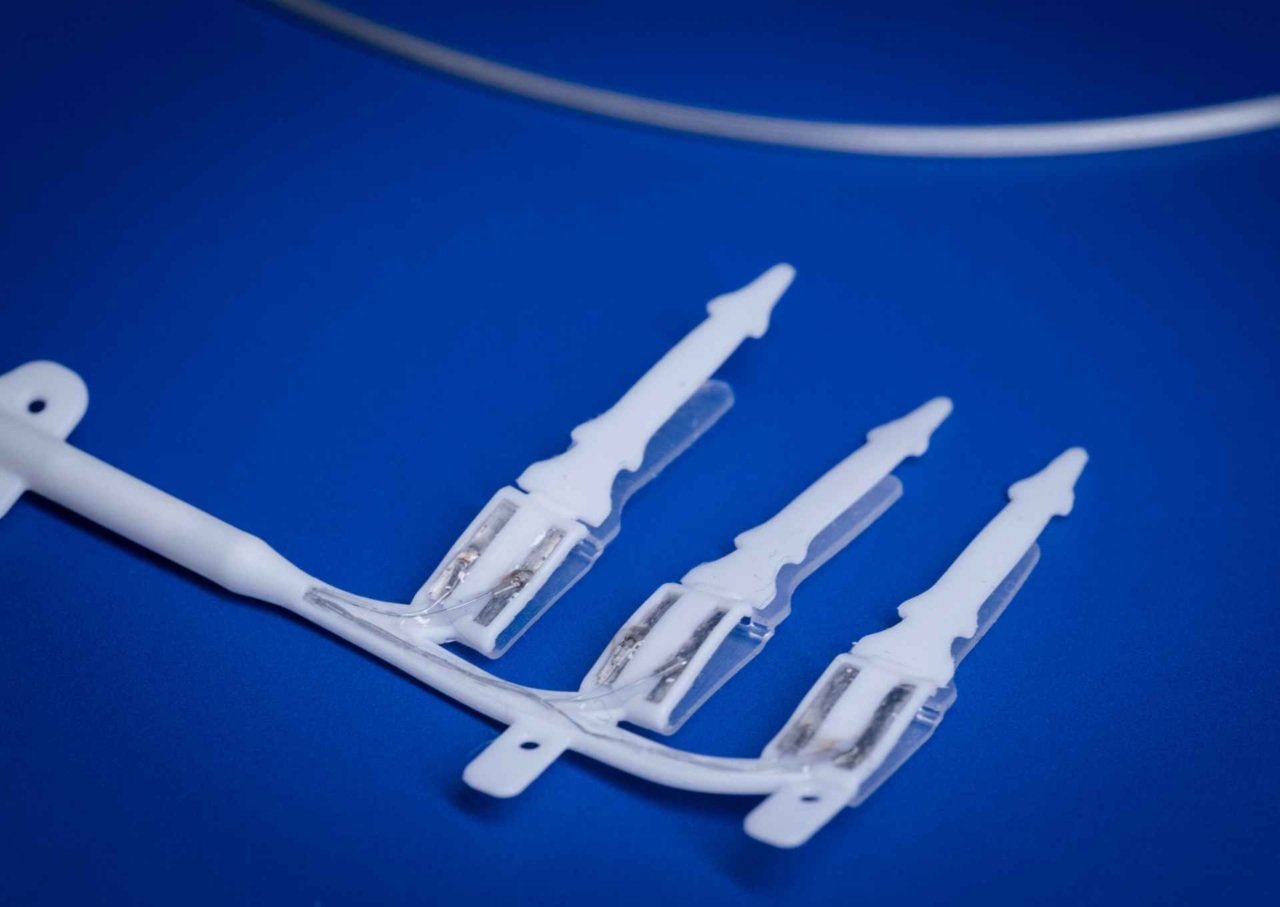
“We have designed the device to be a set-and-forget treatment powered by a small battery under the skin, hopefully allowing patients to continue
their lives without the fear of further surgery.”
“Up to 80 per cent of Crohn’s sufferers will require surgery at some point in their lives. It’s hoped that eventually this new device will work as a set and forget, allowing patients to continue with their lives without the fear of ongoing treatment with medication,” said Associate Professor Peter De Cruz.
If successful, this study will confirm safety and efficacy of the device for Crohn’s sufferers and provide data to understand if remission can be sustained after surgery.