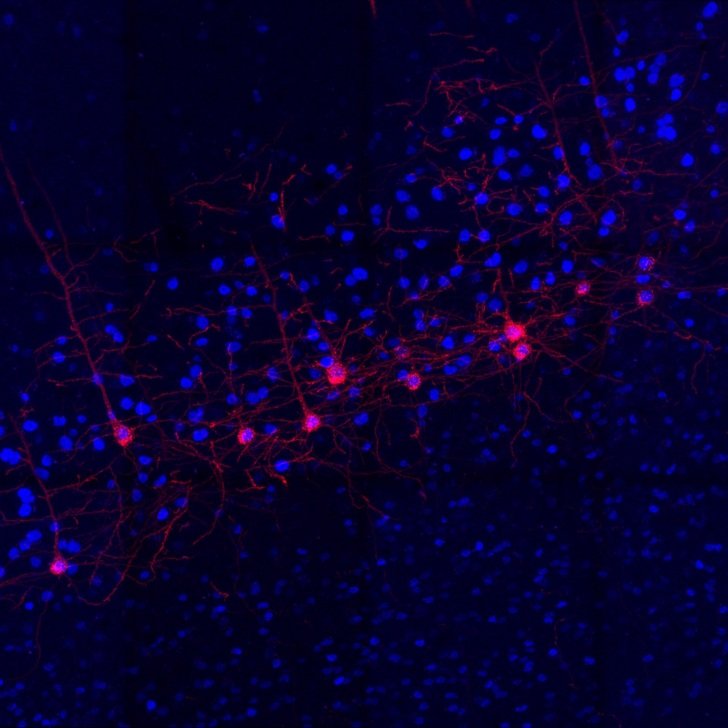
Research by Florey PhD student, Rachel Peiris, has revealed the effects of blocking signaling from the splanchnic sympathetic nerve – nerves that control the functions of the gut and pelvic organs – during sepsis may pave the way forward for rapidly clearing live bacteria from the bloodstream during sepsis. The work has been published in Intensive Care Medicine Experimental.
Rachel’s current testing on preclinical models has shown that cutting this nerve at 24 hours of established sepsis did not have any adverse effects on the cardiovascular function.
Importantly it showed that by 26 hours of infection a clinically relevant time point when septic patients are usually treated in ICU, there was a reduction of bacteria in the blood after just 2 hours of denervation – when the nerves were cut.
“We were able to show that it is safe for blood pressure and heart rate control, and it helped to promote an immune response that resulted in a faster resolution of the bacterial infection. This was followed by a sustained improvement in the behavioral state of the model,” Rachel said.
The recent published findings follow research dating back to 2020, when Florey researchers found that when the splanchnic nerves are cut on both sides in a preclinical model, the inflammatory response to live bacteria (E. coli) was activated which led to the rapid clearance of bacteria from the bloodstream within 1.5 hours.

Sepsis occurs when there is an excessive immune and inflammatory response to an infection.
It is the leading cause of death in intensive care units and despite current treatments and it kills nearly 11 million people every year worldwide. There is currently no treatment to reverse sepsis-induced multi-organ failure.
During sepsis, there are two phases: a heightened level of immuno-activation within the first 24 hours, meaning your immune cells have identified a pathogen and are seeking to find it. But after that period of time, the body goes into immuno-suppression, a protracted phase that lasts up to weeks.
During this phase is when patients are immuno-paralysed, immune cells are not fighting the infection and can lead to hypotension (low blood pressure), multiple organ dysfunction and death.
“Sepsis is one of the biggest causes of death in the ICU. Our work is successfully paving a way forward to prevent extreme illness and in many cases death,” Rachel said.




![[MAY] Neuroscope – Resizing (500 x 500 px)](https://florey.edu.au/wp-content/uploads/2026/03/MAY-Neuroscope-Resizing-500-x-500-px-.png)