Researchers at The Florey will lead a world-first clinical trial investigating ambroxol as a treatment for motor neurone disease (MND). The trial will assess the safety and efficacy of the treatment thanks to $2 million in funding awarded by FightMND.
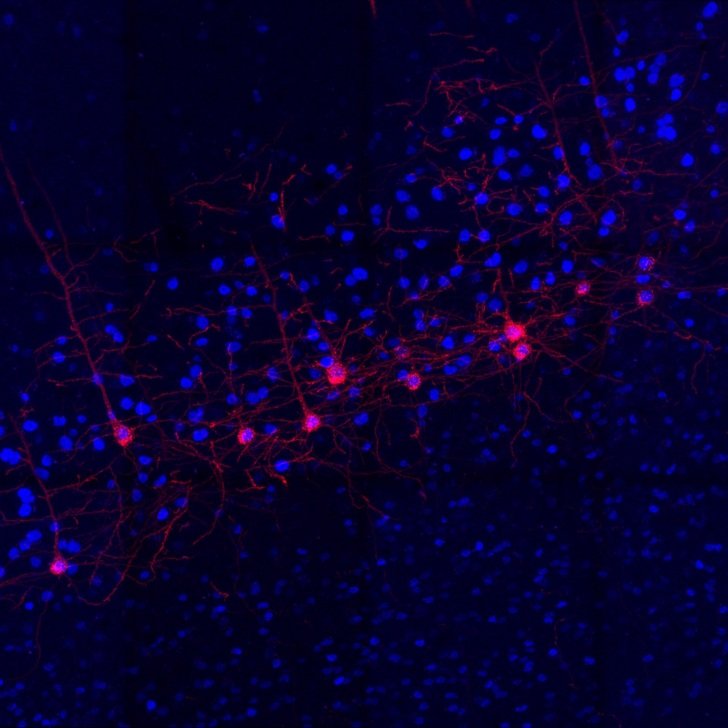
Head of the MND Laboratory at The Florey, Professor Brad Turner, will lead an international team to repurpose existing drug ambroxol, a safe prescription medication used as a cough suppressant.
This will evaluate the drug as a possible treatment in MND for the first time.
“Ambroxol is an ideal drug candidate to assess in MND research. It is a safe drug – orally administered as a syrup, it enters the brain and targets multiple key disease pathways implicated in MND, including the disruption of connections between the brain and muscles that occurs in MND,” said Prof Turner.

The team hopes to build on earlier research that found ambroxol builds stronger connections between the nervous system and muscles.
“Previously we revealed a novel action of ambroxol in correcting abnormal metabolism of fat molecules [known as ‘lipids’] that contribute to the breakdown of communication between motor neuron function and muscle leading to disease progression,” he said.
The team demonstrated correcting abnormal lipid metabolism in animal models of MND where they saw a slowing of both disease progression and better protection of motor neurons and their connections to muscle. This is the first time that drug efficacy has been independently replicated across three distinct animal models of MND.
The team now hope to replicate this for patients with MND.
The two-year trial is recruiting people with MND across the country and treating them with escalating oral doses of ambroxol for 6-months. These data will enable the team to define an effective dose of ambroxol which they can then use to take the therapy to larger trials and better establish if the drug is disease-modifying in MND.
Professor Turner said the project had been many years in the making.
This phase two clinical trial builds upon many years of research made possible from our ongoing partnership and the generous funding received from FightMND and its supporters.
The trial will be conducted in collaboration with clinical investigators Professor Steve Vucic of the Brain and Nerve Research Centre, Concord Repatriation General Hospital, and Professor Matthew Kiernan of the Brain and Mind Centre, University of Sydney.





![[MAY] Neuroscope – Resizing (500 x 500 px)](https://florey.edu.au/wp-content/uploads/2026/03/MAY-Neuroscope-Resizing-500-x-500-px-.png)