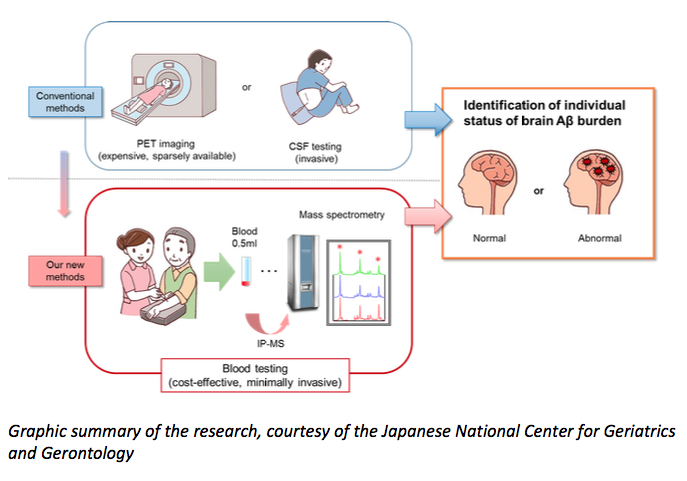
The blood test measures a specific peptide in the blood to inform scientists, with 90 per cent accuracy, if a patient has the very earliest stages of Alzheimer’s disease.
Blood samples from patients in a large study from the Japanese National Center for Geriatrics and Gerontology (NCGG) were initially analysed to identify the relevant peptides. Those indicating brain beta-amyloid burden were then tested against patient samples from the Australian Imaging, Biomarker and Lifestyle Study of Aging (AIBL), to validate the results.
Professor Katsuhiko Yanagisawa, Director-general of Research Institute at NCGG, says: “Our study demonstrates the high accuracy, reliability and reproducibility of this blood test, as it was successfully validated in two independent large datasets from Japan and Australia.”
Dr Koichi Tanaka at Shimadzu Corporation was instrumental in developing the initial blood testing procedure. Professor Tanaka won the Nobel prize in Chemistry in 2002 for the technique. “From a tiny blood sample, our method can measure several amyloid-related proteins, even though their concentration is extremely low. We found that the ratio of these proteins was an accurate surrogate for brain amyloid burden.”
One of the essential hallmarks of Alzheimer’s disease is buildup of abnormal peptide in the brain, called beta-amyloid. The process starts silently about 30 years before outward signs of dementia, like memory loss or cognitive decline, have begun.
Current tests for beta-amyloid include brain scans with costly radioactive tracers, or analysing spinal fluid taken via a lumbar puncture. These are expensive and invasive, and generally only available in a research setting. A diagnosis is usually made without these tools, by assessing a patient’s range of symptoms.
Laureate Professor Colin Masters of the Florey Institute of Neuroscience and Mental Health, and The University of Melbourne, has been at the forefront of Alzheimer’s research since the 1980s. Professor Masters, who co-led the research published in the latest issue of Nature, says, “This new test has the potential to eventually disrupt the expensive and invasive scanning and spinal fluid technologies. In the first instance, however, it will be an invaluable tool in increasing the speed of screening potential patients for new drug trials.”
“Progress in developing new therapeutic strategies for Alzheimer’s disease has been disappointingly slow. None of the three drugs currently on the market treat the underlying disease.
New drugs are urgently required, and the only way to do that is to speed up the whole process. That requires trials with rigorous and economical patient selection, to avoid recruiting patients who may not even have Alzheimer’s disease.
Due to the long timespans involved, pharmaceutical companies require accurate predictions of who is most at risk.”
The breakthrough was the result of an extensive international collaboration between industry and academia in both Japan and Australia. The key technology, known as IP-MS, was implemented by Shimadzu Corporation in Japan. The University of Tokyo, Kyoto University, Kindai University, and Tokyo Metropolitan Institute of Gerontology all contributed to the work. In Australia, the AIBL study upon which the test was validated, is a partnership between the Florey Institute, The University of Melbourne, CSIRO, Edith Cowan University, and Austin Health.
The research is available to view on the Nature website.
